Brand vs Generic Medicine: Are Generic Medicines Really as Effective?
Please note: This article gives basic advice only. If you are experiencing an issue, please don’t self-diagnose. Talk to one of our pharmacists online — we’ll help you find out what’s really going on and what medicine or supplement is safest for you.
Contents
- Introduction
- Overview of brand vs generic medicines
- Regulatory context in Nigeria (NAFDAC)
- What is the difference between a brand medicine and a generic medicine?
- Are generic medicines as effective as branded ones?
- Bioequivalence explained
- Regulatory standards (FDA & NAFDAC)
- When caution is needed
- Why generic medicines are cheaper
- Development cost differences
- Market competition
- Real-life affordability impact in Nigeria
- Are generic medicines examined thoroughly in Nigeria?
- NAFDAC regulations and updates
- Bioequivalence requirements
- Scientific review process
- What is bioequivalence in simple language?
- How pharmacists choose generic substitutes
- Active ingredient
- Strength and dosage form
- Route of administration
- Regulatory approval
- Patient condition
- Patient history
- When might extra caution be needed?
- Narrow therapeutic index medicines
- Modified-release medicines
- Stable long-term treatments
- Sensitivity to inactive ingredients
- Common myths about generic medicines
- Myth 1: Generics are fake
- Myth 2: Generics are weaker
- Myth 3: Generics are poor quality
- Myth 4: Any generic can replace any brand
- Practical advice for patients
- Key questions to ask when switching
- Final takeaway
- Sources
Introduction
When patients see a familiar brand replaced with a lower-cost generic, it is normal to have questions. Are generics really as effective? Why is the price so different? And how do pharmacists decide when a generic substitute is appropriate?
The good news is that a properly approved generic medicine is meant to work the same way as its brand-name version. In Nigeria, NAFDAC has also strengthened its bioequivalence framework, including updated guidance in 2024 and 2025 and a 2025 public briefing that says bioequivalence establishes the interchangeability of generic medicines with branded equivalents. NAFDAC’s 2025 registration guidance for drug products also says generic products must submit a bioequivalence study report or biowaiver data as part of registration. (NAFDAC)
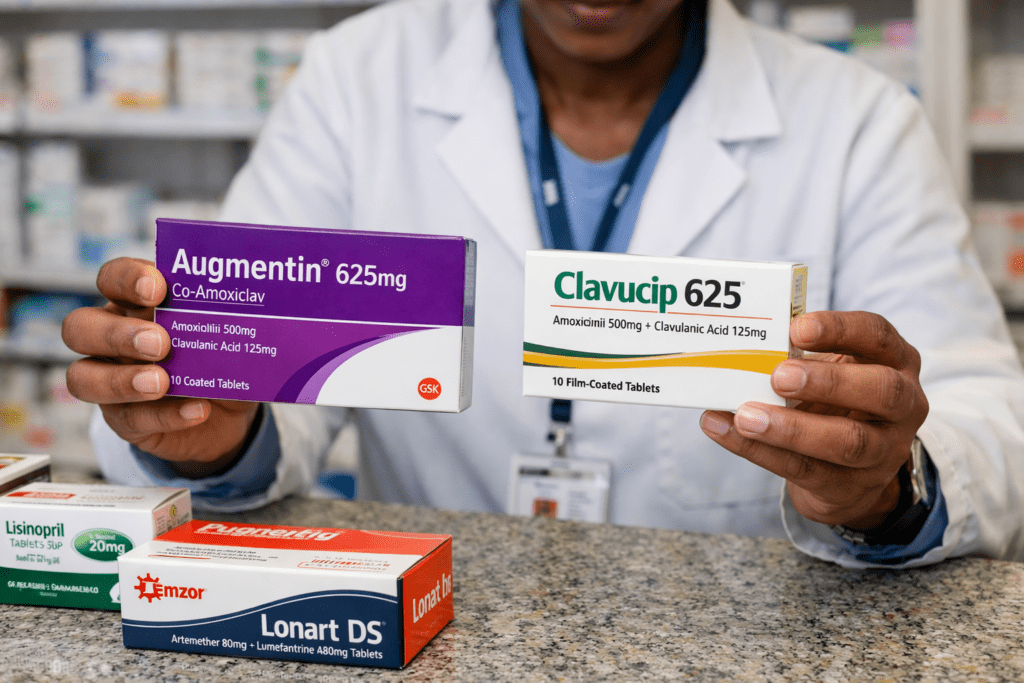
What is the difference between a brand medicine and a generic medicine?
A brand medicine is the original product developed and first marketed by a company under a trade name. A generic medicine is made to match that original medicine after patent or exclusivity periods allow other manufacturers to produce it. A generic is expected to have the same active ingredient, strength, dosage form, route of use, and intended effect, though the colour, shape, packaging, or some inactive ingredients may differ. (U.S. Food and Drug Administration)
So, if the active treatment ingredient is the same and the product meets regulatory standards, the medicine should give the same clinical benefit for most patients. (U.S. Food and Drug Administration)
Are generic medicines as effective as branded ones?
In general, yes. Properly approved generic medicines are expected to be as effective as branded ones. Regulators require evidence that the generic performs the same way in the body as the brand medicine. This is called bioequivalence. (U.S. Food and Drug Administration)
The U.S. FDA explains that a generic medicine must be the same as the brand medicine in dosage form, safety, strength, route of administration, quality, performance characteristics, and intended use, and must provide the same clinical benefit. The FDA also notes that small batch-to-batch differences can happen with both brand and generic medicines, but only within acceptable limits. (U.S. Food and Drug Administration)
NAFDAC’s own guidance says bioequivalence studies are an essential part of the regulatory process for approval of generic medicines, and that the results provide the basis for determining therapeutic equivalence and interchangeability with the reference product. (NAFDAC)
That said, “as effective” does not mean every switch should be done casually. Some medicines need extra care, especially where small differences in blood levels can matter more, such as certain modified-release medicines or medicines with a narrow therapeutic index. Pharmacists consider this before recommending a substitute. (PMC)
Why generic medicines are cheaper
One of the biggest reasons patients doubt generics is price. People often assume: “Cheaper means weaker” , but that is usually not the reason.
Generic medicines are often cheaper because the manufacturer does not have to repeat the original large animal and human studies that were used to prove the medicine’s safety and effectiveness when it was first developed. Instead, the generic maker must show that its product matches the approved reference medicine closely enough to be substituted. Also, once multiple generic companies enter the market, competition usually drives prices down. (U.S. Food and Drug Administration)
The NHS explains it simply: generic medicines contain the same active ingredients as the branded medicine and are used more often because they are just as effective but cost far less. (nhs.uk)
This matters in everyday life. In Nigeria, where many patients still pay out of pocket for medicines, lower-cost generic options can make treatment more affordable and improve the chance that patients actually continue their treatment instead of stopping midway because of cost. Older Nigerian research found that innovator brands could cost 2 to 7 times more than the lowest-priced generics, and affordability remains a major real-world issue. (PMC)
Are generic medicines examined thoroughly in Nigeria?
This is the key concern, and the answer is: Nigeria’s regulators do examine them, and the requirements have become stricter.
NAFDAC has published guidance on establishing interchangeability of generic pharmaceutical products, stating that the purpose is to assure interchangeability without compromising safety, efficacy, and quality. (NAFDAC)
More recently, NAFDAC updated its bioequivalence guidance in 2024 and 2025. Its April 10, 2025 press briefing said the framework establishes the interchangeability of generic medicines with branded equivalents using statistically significant clinical evidence, and NAFDAC’s registration guidance in 2025 states that for generic products, a bioequivalence study report or biowaiver data must be submitted in the Common Technical Document dossier format. (NAFDAC)
NAFDAC also states that building Nigeria’s bioequivalence ecosystem will help ensure the quality, safety, and efficacy of generic medicines in the country. (NAFDAC)
So, based on current regulator guidance, Nigeria’s health sector does not treat generics as casual copies. They are expected to go through a scientific review process aimed at proving they can be used interchangeably with the brand product. (NAFDAC)
What is bioequivalence in simple language?
Bioequivalence means the generic medicine gets into the body in a way that is close enough to the brand medicine that it should work the same in practice. It acts as a scientific bridge between the original product, which already has clinical effectiveness data, and the generic version. (WHO Extranet)
This does not mean the tablet must look the same. It may have a different colour, shape, or filler ingredients. What matters is that the active ingredient and performance are shown to match the approved standard closely enough. (U.S. Food and Drug Administration)
How pharmacists choose generic substitutes
Pharmacists do not pick generic substitutes at random.
A careful pharmacist usually checks several things before recommending a switch:
1. The active ingredient
The generic must contain the same active ingredient as the prescribed brand. (U.S. Food and Drug Administration)
2. The same strength and dosage form
A 500 mg tablet should be replaced with a 500 mg tablet, not a syrup or a different strength unless the prescriber intentionally changes it. (U.S. Food and Drug Administration)
3. The same route of administration
An oral tablet should be substituted with an oral product, not an injection or cream. (U.S. Food and Drug Administration)
4. Regulatory approval
Pharmacists look for products that are properly registered and sourced through legitimate supply channels. In Nigeria, NAFDAC regulates medicines, while the Pharmacy Council of Nigeria regulates pharmacy practice and standards. (NAFDAC)
5. The patient’s condition
Not every medicine switch is appropriate in every situation. Research among pharmacists in Nigeria found strong support for generic substitution, but also agreement that substitution is not appropriate in all situations, especially for certain sensitive medicines. (PMC)
6. The patient’s past experience
If a patient has previously reacted badly to a specific formulation, had confusion because of repeated brand changes, or struggles with adherence, the pharmacist may be more cautious. Nigerian pharmacists in one study reported that generic substitution sometimes requires more patient counselling and follow-up. (PMC)
When might extra caution be needed?
Even though generics are usually appropriate, some situations deserve closer review:
- Medicines with a narrow therapeutic index
- Modified-release or controlled-release products
- Patients who are already stable on a long-term medicine and may become confused by frequent switching
- Cases where a patient may react to a specific inactive ingredient rather than the active drug itself (PMC)
This is why pharmacist advice matters. The right answer is not always “brand only” or “generic only.” The right answer is: use the safe, approved option that fits the patient’s condition, budget, and treatment plan.
Common myths about generic medicines
Myth 1: “Generic medicines are fake”
False. A genuine generic medicine is a legitimate medicine, not a counterfeit. Counterfeit medicines are illegal fake products. A proper generic is an approved version of the same medicine. (NAFDAC)
Myth 2: “Generic drugs are weaker”
False in principle. Approved generics are expected to provide the same clinical benefit as the brand version. (U.S. Food and Drug Administration)
Myth 3: “They are cheaper because they are poor quality”
Usually false. They are often cheaper because development costs are lower and competition is stronger after patents expire. (U.S. Food and Drug Administration)
Myth 4: “Any generic can replace any brand in every situation”
Not always. Some substitutions need more caution, and pharmacist or prescriber review may be needed. (PMC)
Practical advice for patients
If you are offered a generic substitute, ask:
- What is the active ingredient?
- Is it the same strength?
- How should I take it?
- Is it okay to switch from my current brand?
- Will the appearance be different?
- What should I watch out for after switching?
These are good questions, not troublesome questions.
A good pharmacist should be able to explain why the substitute is appropriate and what differences matter and do not matter.
Final takeaway
For most common medicines, a properly approved generic medicine should work just as well as the branded version. The lower price usually reflects lower development costs and stronger market competition, not lower treatment value.
Still, medicine substitution should be done thoughtfully. That is where pharmacists are especially important: checking the active ingredient, strength, dosage form, safety, patient history, and whether a switch makes sense in real life.
If you are unsure whether to choose a brand or a generic medicine, speak with our pharmacists online. We can help you compare options, avoid confusion, and choose the safest and most cost-effective treatment for your needs.
Sources
- NAFDAC, Guideline on Registration Requirements to Establish Interchangeability of Generic Pharmaceutical Products. (NAFDAC)
- NAFDAC, Guidelines on the Investigation of Bioequivalence (2024/2025 search records). (NAFDAC)
- NAFDAC, Press Briefing on the Regulatory Framework for Bioequivalence and its Relevance to the Nigerian Market (April 10, 2025). (NAFDAC)
- NAFDAC, Bioequivalence Study in Nigeria. (NAFDAC)
- NAFDAC, Guidelines for Registration of Drug Products Made in Nigeria (2025 search record). (NAFDAC)
- Pharmacy Council of Nigeria official website. (Pharmacy Council of Nigeria)
- U.S. FDA, Generic Drugs: Questions & Answers. (U.S. Food and Drug Administration)
- U.S. FDA, Generic Drug Facts. (U.S. Food and Drug Administration)
- NHS, Medicines information. (nhs.uk)
- WHO, Bioequivalence | Prequalification of Medical Products. (WHO Extranet)
- Auta A. et al., Generic Medicine Substitution: A Cross-Sectional Survey of the Perception of Pharmacists in North-Central, Nigeria. (PMC)
You may also like
- Brand vs Generic Medicine: Are Generic Medicines Really as Effective?
- A Practical Guide to Weight Management, Weight Loss, and Nutrition
- A Guide to Preventive Health & Everyday Wellness
- Women’s Health, Made Simple: A Simple Guide for Healthy Living
- Men’s Health, Made Simple: A Guide to Feeling and Living Better